Background
V. cholerae requires the enterotoxin cholera toxin (CT) to cause severe diarrheal disease. The catalytic activity of CT activates adenylyl cyclase, which increases cellular levels of 3′,5′-cyclic AMP (cAMP) in target cells, activating of the cAMP-dependent protein kinase PKA. CT is also thought to promote transmission of the pathogen, as infected individuals shed many liters of diarrheal fluid typically containing over 1011 organisms per liter. CT-mediated activation of PKA also leads to upregulation of Hormone Sensitive Lipase (HSL), which catalyzes the lipolysis of triglycerides in adipose cells and the release of free long-chain fatty acids (LCFAs) and glycerol. The CT-mediated increase in cAMP can also induce a metabolic switch to anaerobic glycolysis in cells, leading to reduced consumption of oxygen and increased production of L-Lactate. We recently found that CT-induced disease leads to increased luminal concentrations of L-lactate, LCFAs metabolites and LCFA uptake-mediated growth of cholerae in the lumen of the small intestine. However, the molecular mechanisms by which CT modulates host-pathogen metabolism and pathogen growth are not fully understood.
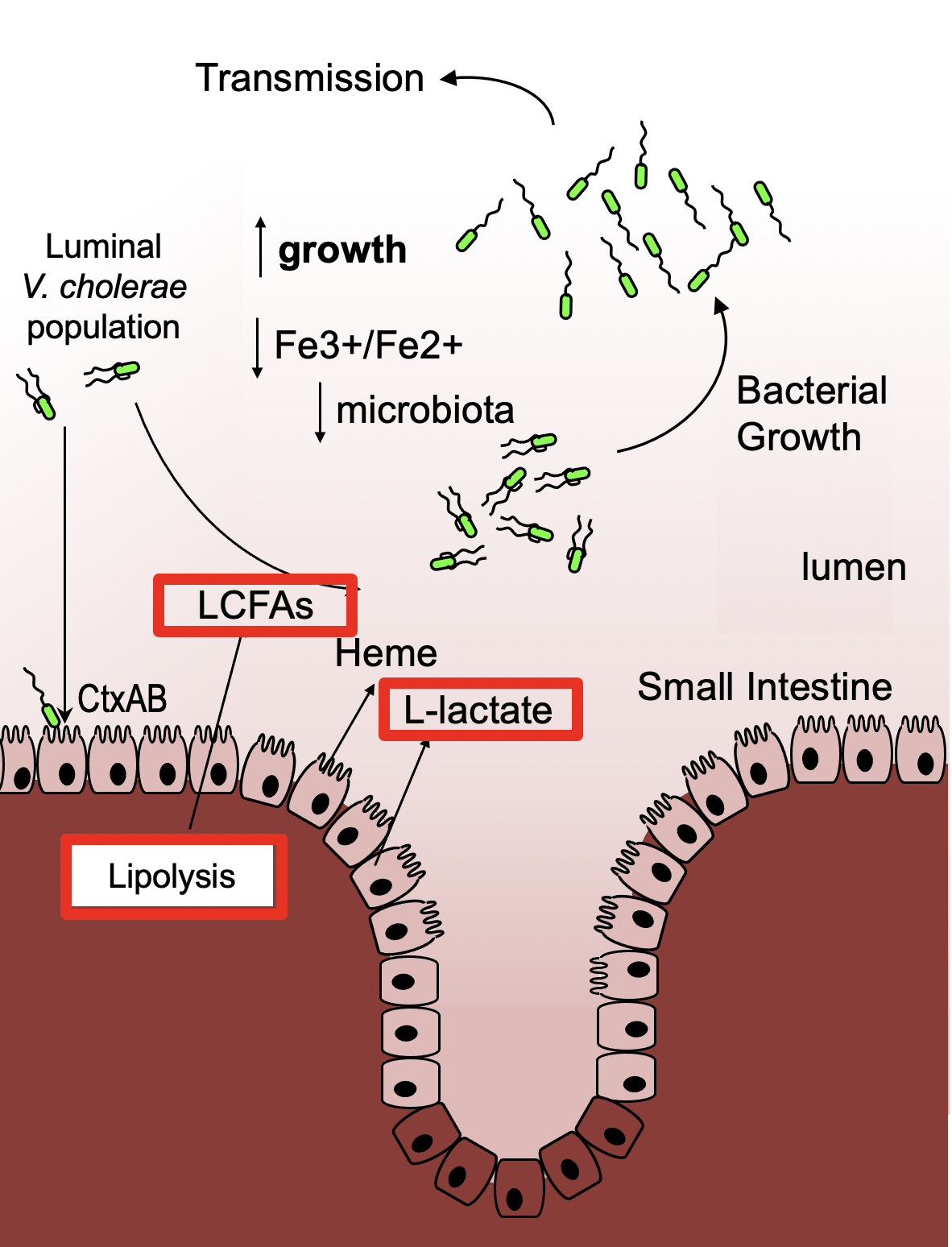
Modulation of Host-Pathogen Metabolism
The infant rabbit model of V. cholerae infection recapitulates many of the CT-dependent disease symptoms observed in humans. Neonatal (suckling) mice also exhibit CT-dependent diarrheal disease and are commonly used as a model for evaluating intestinal colonization by V. cholerae. We utilize both animal models to study the molecular mechanisms of toxin-induced modulation of host-microbe metabolism, pathogen growth, and transmission. During his postdoctoral research, Dr. Rivera-Chávez found that cholera toxin produced by Vibrio cholerae during infection, alters host intestinal metabolism and provides the pathogen with a novel nutrient niche for growth (Rivera-Chavez et al, 2019). We will utilize our tools to examine how cholera toxin and other bacterial toxins modulate both host and pathogen metabolism during disease (See Figure). These findings will have broad implications for how CT and other bacterial toxins modulate host-pathogen metabolism and pathogen growth, which could lead to novel therapeutics and intervention strategies.


